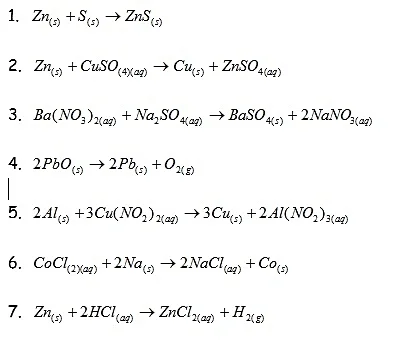Subject: Chemistry
Topic: Chemical Reactions
Time Frame: 60 –75 minutes
Objectives:
- Students will be able to identify the different types of chemical reactions
- Students will examine the chemical reactions used for water treatment
Methodology:
- Demonstration
- Independent study
- Direct Lecture
Materials:
- Student worksheet
- Lecture notes · Beakers
- Test tube
- Bunsen Burner
- Matches
- Calcium chloride solution
- Sodium carbonate solution
- HCl
- Zinc metal
- Copper Sulfate Pentahydrate
- Copper Sulfate Anhydrous
Space Requirements:
- There should be enough space to allow the teacher to safely demonstrate the chemical reactions.
Background Information:
- Students should have a basic understanding of what a chemical reaction is. As well, students should be able to write and name compounds.
Directions/Procedure:
- Explain to the students that chemical reactions occur every day in our lives. Chemical reactions even help to provide clean water for us to drink.
- Demonstrate to the students the different types of chemical reactions. Use this demonstration as well to review the signs that a chemical reaction has occurred (colour change, precipitation formed, gas formed and heat released).
a. Hand out the observation sheet for the students to fill out as you demonstrate the equations. Explain to the students that they will not be able to fill out the Type of Reaction column yet.
b. Mix together 10ml of calcium chloride and 10 mL of sodium carbonate. Ask the students to write down what they observe and the chemicals that you used.
c. Put 20mL of HCL into a beaker and add a small piece of zinc.
d. Take a spatula full of copper sulfate pentahydrate and put it into a test tube. Slowly heat the test tube using a Bunsen burner until an observation can be made.
e. Take a scoop full of copper sulfate anhydrous and put it into a beaker. Add 5mL of water.
- As a class, ensure that the students have correctly written down the reactants of each reaction.
- Explain to the students that each demonstration was a different type of reaction. Ask the students what they noticed was anything different between each of the reactions.
- Direct Lecture
Explain the difference between a combination, decomposition, single displacement and double displacement reaction.
- Ask the students to fill out the Type of Reaction column on their observation sheet.
- Hand out the student worksheet to further ensure the students are able to differentiate between the different types of reactions. Allow the students 10 minutes to complete this.
- Direct Lecture
a. Explain to the students that there are different types of reactions needed to help ensure our water is safe to drink.
i. The most common reaction occurs when chlorine is added to the water to kill the bacteria and viruses in the water.
ii. Lime (calcium hydroxide) reacts with water to remove iron and carbon dioxide
iii. Hypochlorite salts are added to water to help disinfect it before it is distributed.
iv. Powdered activated carbon is added to water to remove dissolved organic materials
(These are just some sample reactions that occur during the water treatment process)
b. Explain to the students that different types of chemical reactions are needed in the water treatment process in order to get rid of the different type of impurities found in water.
- Closure – Ask the students to find out if their parents do any treatment to their water at home (i.e. well water, water softening).
Evaluation:
Formal Assessment – Marking of the worksheet
Informal Assessment – Observation of the students’ behaviour during the demonstrations
Resources:
Safe Drinking Water Foundation
www.safewater.org
Drinking Water and Groundwater Kids' Stuff
https://www3.epa.gov/safewater/kids/index.html
Types of Reactions Worksheet
Classify the following reactions as decomposition, combination, single displacement or double displacement.